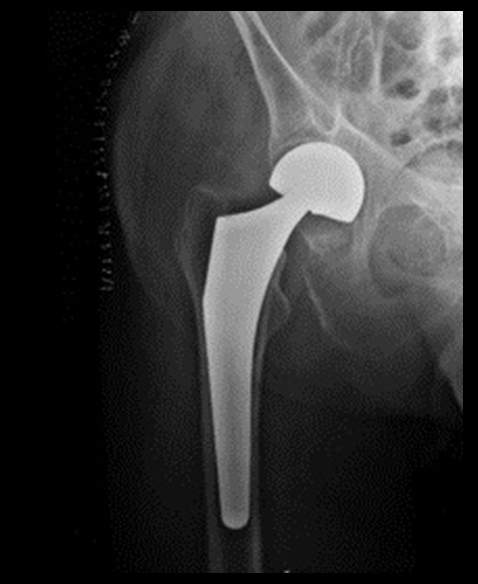
In 2012, Stryker Corp. recalled two of its hip replacement systems – the Rejuvenate and ABG ll. In all, about 20,000 people were implanted with one of those systems. Among the dangers associated with these devices are adverse reactions resulting from large quantities of metal entering a patient’s bloodstream and tissue.
Thousands of people across the country have filed lawsuits against Stryker Corp. for damage caused by the two recalled hip replacement systems. A recent settlement covers around 3,000 patients in 39 states who already had revision surgery to remove the implant before Nov. 3, 2014. The settlement is ‘uncapped’ as Stryker agreed to pay at least $1 billion, but total payouts are estimated to be much higher. In fact, Stryker announced that it had set aside more than $1.4 billion. Patients deemed by their surgeon to be physically unable to undergo the revision procedure to remove the recalled implant may also be eligible to make a claim against the settlement. The claims and lawsuits of patients whose artificial hips were not replaced before Nov. 3, 2014 are not affected by this settlement, but may be covered by a future settlement. For more information, click here for the official settlement program website.
It is critical that patients with Stryker hip replacement products quickly consult their surgeon to determine whether they have one of these recalled devices, whether there are signs of the adverse reactions, and whether the device needs to be removed.